Original article
Safety and feasibility of photobiomodulation (LASER acupuncture) for pain in children with sickle cell disease: a pilot study
Carla Verônica Paixão Marques¹ (https://orcid.org/0000-0001-7961-3982)
Isadora Nunes Sá Fontes Baptista² (https://orcid.org/0000-0002-7272-4978)
Ana Marice Teixeira Ladeia³ (https://orcid.org/0000-0002-2235-7401)
André Fonseca⁴ (https://orcid.org/0000-0003-1573-3058)
Katia Nunes Sá⁵ (https://orcid.org/0000-0002-0255-4379)
Abrahão Fontes Baptista⁶ (https://orcid.org/0000-0001-7870-3820)
1Universidade Salvador (Salvador). Bahia, Brazil. Maternidade Maria da Conceição de Jesus (Salvador). Bahia, Brazil.
2Instituto Lume para Desenvolvimento Humano (Petrópolis). Rio de Janeiro, Brazil. Núcleo de Assistência e Pesquisa em Neuromodulação – NAPeN network (Recife). Pernambuco, Brazil.
3Escola Bahiana de Medicina e Saúde Pública (Salvador). Bahia, Brazil.
4Universidade Federal do ABC (Santo André). São Paulo, Brazil.
5Corresponding contact. Escola Bahiana de Medicina e Saúde Pública (Salvador). Bahia, Brazil. Núcleo de Assistência e Pesquisa em Neuromodulação – NAPeN network (Recife). Pernambuco, Brazil. [email protected]
6Universidade Federal do Rio de Janeiro (Rio de Janeiro). Rio de Janeiro, Brazil. Núcleo de Assistência e Pesquisa em Neuromodulação – NAPeN network (Recife). Pernambuco, Brazil.
ABSTRACT | INTRODUCTION: Sickle cell disease (SCD) presents pain as one of its primary symptoms, manifesting from early childhood, and requiring effective strategies for its control and management. OBJECTIVE: To evaluate the feasibility, safety and preliminary effects of photobiomodulation (LASER acupuncture) for pain control in children with sickle cell disease. Secondly, we decided to analyze the changes in brain waves before and after a single session. METHOD: A pilot study was conducted in children aged 6 to 12 years, followed at the Pediatric Hematology Outpatient Clinic of Professor Edgard Santos University Hospital, Federal University of Bahia (Universidade Federal da Bahia - UFBA), Brazil. A body diagram, the Wong-Baker Faces Pain Rating Scale, the PedsQL Quality of Life Questionnaire, and electroencephalographic (EEG) recordings were applied before and after a single session of acupuncture through LASER therapy photobiomodulation based on Traditional Chinese Medicine procedures. The brain activities were evaluated comparing children with SCD with healthy children paired by age and sex. RESULTS: Ten girls and five boys participated in the study, with a mean age of 9.93 ± 1.71 years. Most (80.00%) had HbSS phenotype, with pain primarily located in the lumbosacral region, knees, and head. The mean pain intensity was 6.00 ± 1.633, described as severe (93.3%), involving sensory, affective, and mixed components. A positive immediate effect was observed with photobiomodulation, reducing pain intensity to 1.75 ± 2.363 (p = 0.036). Quantitative EEG tracings also revealed in an exploratory analysis a modulation potential for alpha, theta, beta, gamma, and sensorimotor waves. CONCLUSION: Acupuncture using LASER therapy photobiomodulation demonstrates promising potential for brain-mediated pain modulation in children with sickle cell disease.
KEYWORDS: Sickle Cell Disease. Pain. Central Sensitization. Photobiomodulation. Acupuncture Therapy.
How to cite this article: Marques CVP, Baptista INSF, Ladeia AMT, Fonseca A, Sá KN, Baptista AF. Safety and feasibility of photobiomodulation (LASER acupuncture) for pain in children with sickle cell disease: a pilot study. Brain Imaging Stimul. 2026;5:e6733. https://doi.org/10.17267/2965-3738bis.2026.e6733
Submitted Feb. 3, 2026, Accepted Mar. 26, 2026, Published May 7, 2026
Brain Imaging Stimul., Salvador, 2026;5:e6733
https://doi.org/10.17267/2965-3738bis.2026.e6733
ISSN: 2965-3738
Assigned editors: Maruo Muskat, Elisabete Carrara
1. Introduction
Sickle cell disease (SCD) is an inherited condition caused by a mutation in the β-globin gene and is highly prevalent in Brazil1. Among its primary symptoms, fatigue and chronic pain manifest from early childhood in individuals with SCD and are considered determining factors for suffering, as well as diminished quality of life and life expectancy among those affected2. As a neglected disease that predominantly affects people of Afro-descendant, SCD is also socially stigmatized3. To minimize morbidity, reduce premature mortality, and maximize quality of life, care measures must be implemented as early as possible.
In addition to well-known biological causes — such as vaso-occlusive crises, hemolytic anemia, splenic sequestration, ischemic strokes, and recurrent infections — which contribute to multi-organ failure and decreased functionality, various psychosocial manifestations are also associated with the distress experienced by children with SCD4. It has been suggested that angiotensin receptor blockades are related to the development of mental disorders and chronic pain in this population5. For this reason, it is essential to understand neurophysiological responses, particularly those occurring in the brain, as 39% of children with SCD are affected by silent cerebral infarcts6 and early-onset mental disorders such as depression, anxiety, social isolation, and suicidal ideation7.
Chronic pain (CP) directly impacts emotional reactions, sleep quality, and fatigue. In children with SCD, CP impairs physical, social, and cognitive functions, and especially the capacity for self-regulation. A consensus on SCD treatment emphasizes the importance of assessing pain intensity and its impact on neurocognitive development, emotional well-being, fatigue, occupational functions, and self-efficacy in children with SCD2. Changes in gray matter volume have also been associated with the presence of pain and reduced quality of life in children with SCD8. It is believed that the strong correlation between alterations in brain structure and connectivity involving the cingulate cortex and the pronounced pain experience may be related to a central pain mechanism9.
Central sensitization in CP among children with SCD requires interventions capable of modulating brain plasticity10. A developing brain possesses greater neuroplastic capacity to respond to stimuli, which should be leveraged as a window of opportunity. In this context, LASER acupuncture, a type of photobiomodulation, has emerged as a promising alternative. LASER (an acronym for Light Amplification by Stimulated Emission of Radiation) at low power delivers light energy with therapeutic effects. When applied to appropriate acupuncture points, it can serve as an anti-inflammatory, analgesic, and regenerative therapy that may be useful in managing pain in children with SCD. The effect of photobiomodulation through LASER acupuncture on pain is controversial yet, mainly after a single session11. However, little is known about the potential of this approach to control pain in children with SCD, and the potential influence on brain activity. Hence, the objective of the present study is to evaluate the feasibility, safety and preliminary effects of photobiomodulation (LASER acupuncture) for pain control in children with sickle cell disease. Secondly, we decided to analyze the changes in brain waves before and after a single session.
2. Methods
2.1 Study design and ethical considerations
A pilot study for a posterior randomized, double-blind clinical trial, approved by the Ethics Committee of Professor Edgard Santos University Hospital, Federal University of Bahia (Universidade Federal da Bahia – UFBA) (Opinion No. 3,847,214 and CAAE 65613917.7.0000.0049), was duly registered in REBEC under the number RBR-9jp5sj.
This study was developed as a pilot project for this future clinical trial to assess the feasibility of its implementation, obtain data for sample size calculation, evaluate the safety of the intervention in the target population (since it had never been tested before), and analyze the effect on brain waves that could support the mechanisms of the future intervention in an appropriate, randomized, blinded, and allocation-concealed sample.
2.2 Target population
Children with chronic pain related to sickle cell disease.
2.3 Accessible population
Patients were followed at the Pediatric Hematology Outpatient Clinic of Professor Edgard Santos University Hospital, Federal University of Bahia.
2.4 Inclusion criteria
Individuals aged 6 to 12 years, of any gender, with a confirmed diagnosis of sickle cell disease by sickle cell test or hemoglobin electrophoresis, reporting frequent joint pain, and providing signed informed assent and consent forms.
2.5 Exclusion criteria
History of fracture within the past year or less, osteomyelitis, rheumatic fever, rheumatoid arthritis, malignant leukemia, cerebral palsy, or current use of opioids.
2.6 Assessment instruments
Body diagram for marking painful areas, Wong-Baker FACES® Pain Rating Scale (as per Ordinance No. 9/DGCG of June 14, 2003), the Pain Quality Cards Instrument (ICQD), Pediatric Quality of Life Inventory™ (PedsQL) questionnaire, and quantitative electroencephalogram (qEEG).
2.7 Population and sample
Children with pain associated with SCD were compared to healthy children paired by age and sex. As a pilot study aimed at assessing the feasibility of the clinical trial and intervention safety, the sample size was not predetermined.
2.8 Control group
To evaluate the brain activities, children from this sample were compared to healthy children paired by age and sex who received photobiomodulation through LASER acupuncture on the same points.
In both groups were collected electroencephalogram data. It was not evaluated pain intensity difference between groups because healthy children did not have pain.
2.9 Procedures
Acupuncture sessions were performed with the patient seated, wearing light clothing and protective glasses. During the session, children were asked to remain as quiet and relaxed as possible. The choice of acupoints that conducted the so-called energy treatment according to Traditional Chinese Medicine (TCM) was carried out by observing usual parameters such as pulse, tongue appearance, and psychological and physiological information.
Participating children diagnosed with SCD underwent a 20-minute acupuncture session, with 6 Joules of power in each acupoint planned according to the group in which they were included using Therapy XT/ DMC tool®.
The acupuncture sessions were performed with the patient seated, wearing light clothing and protective eyewear. During the session, the children were asked to remain as quiet and relaxed as possible. The technique was performed by an experienced acupuncturist with 10 years of experience in Traditional Chinese Medicine. The choice of acupuncture points that guided the so-called energy treatment according to Traditional Chinese Medicine (TCM) was made by observing usual parameters, such as pulse, tongue appearance, psychological and physiological information. The choice of the 10 acupuncture points used was made following the Acupuncture Consort12, identifying each individual as unique. The points in this practice were prescribed according to the patient's condition at a given time, but following the choice of meridian points according to the location of the pain through their Luo points, Ashi points, and through the energetic physiology of the pathology which, in the case of SCD, is identified as Xue Deficiency and interrupted Free Flow. The participating children diagnosed with SCD underwent a 20-minute acupuncture session, with 6J of power at each acupuncture point, planned according to the group to which they belong.
After the acupuncture session, the children were asked if they experienced any discomfort, unease, or pain.
2.10 Electroencephalographic evaluation
Visual inspection was made on the first stage, bandpass filters ranging from 0.5 to 40 Hz were used to remove low-frequency noise (baseline deviation) and high-frequency noise (muscle activity), and notch filter rejected 50Hz bands to remove net electrical interferences. Bad channels have been removed, data segmentation into epochs (trials) and elimination of those with very high amplitude (standard deviation-based methods), or interpolation of data from poor channels were performed. The software's own algorithms automatically detected and corrected the artifacts offline.
The relative power density obtained by the electroencephalogram was measured before and after the intervention. An electroencephalograph (EEG Brainet 36, EMSA, Brazil) with 30 electrodes arranged according to the 10/20 electroencephalography system was used in the following electrode locations: F7, T3, T5, Fp1, F3, C3, P3, O1, F8, T4, T6, Fp2, F4, C4, P4, O2, Fz, Cz, Pz, Oz, FT7, FT8, TP7, CP3, FC3, CPz, FCz, CP4, FC4, TP8.
EEG data were collected at a sampling rate of 600 Hz and referenced to the Cz channel. The impedance was kept below 5 KOhm for all electrodes, and the recording environment was maintained with soft lighting and protected by a Faraday cage. Participants were instructed to sit comfortably in a chair, keeping their eyes open while the EEG was recorded for five minutes.
2.11 Data analysis
The collected data were tabulated and analyzed, showing normal distribution. Categorical variables were presented as absolute numbers and proportions, while numerical variables were expressed as means and standard deviations (SD). For the within-group comparison of pain intensity means, a paired Student’s t-test was used. For qEEG tracing evaluation, spectral index analysis was performed, comparing individuals in the AL group before and after the single intervention and with the control group, using the Kruskal-Wallis test. The spectral indexes considered were PRI (the ratio between delta+theta power and alpha+beta power), DAR (the ratio between delta power and alpha power), and TBR (the ratio between theta power and beta power).
3. Results
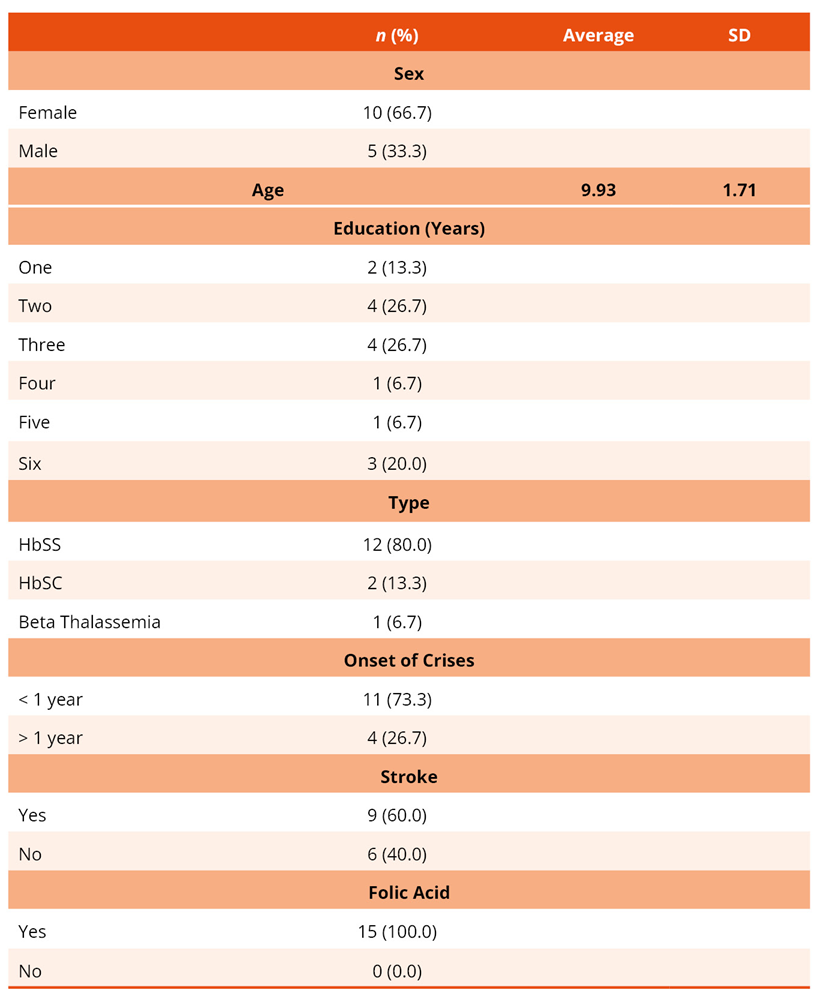
Forty-nine potential participants registered at the reference outpatient clinic were identified. After applying the eligibility criteria, 15 were included in the final pilot study sample. The sociodemographic and clinical characteristics of the participants are presented in Table 1.
Table 1. Sociodemographic and clinical characteristics of the sample of children with sickle cell disease (n = 15)
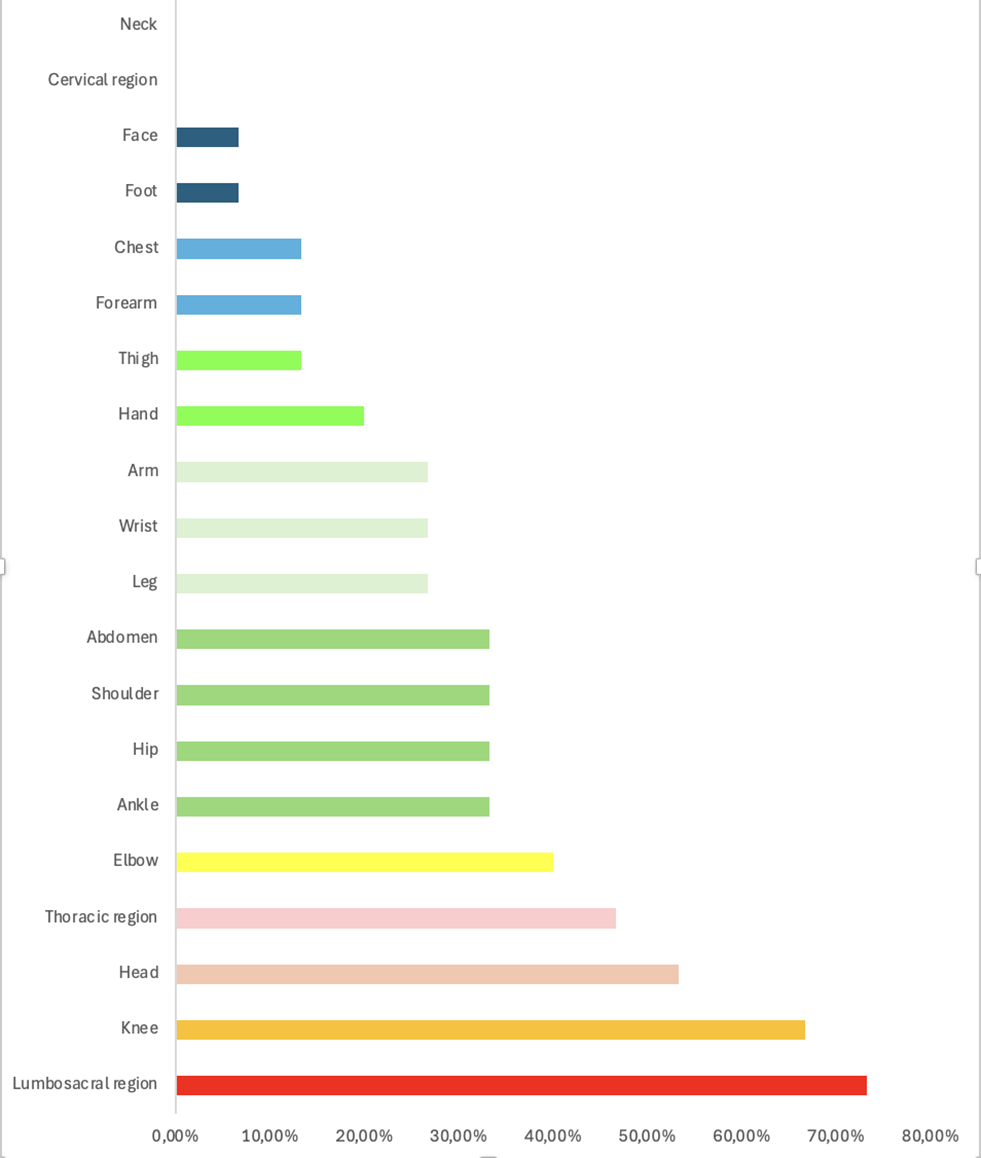
Among the pain sites reported by the participants, the lumbosacral region was the most affected, followed by the knee, head, and thoracic region (Figure 1).
Figure 1. Body regions affected by pain in sample of children with SCD
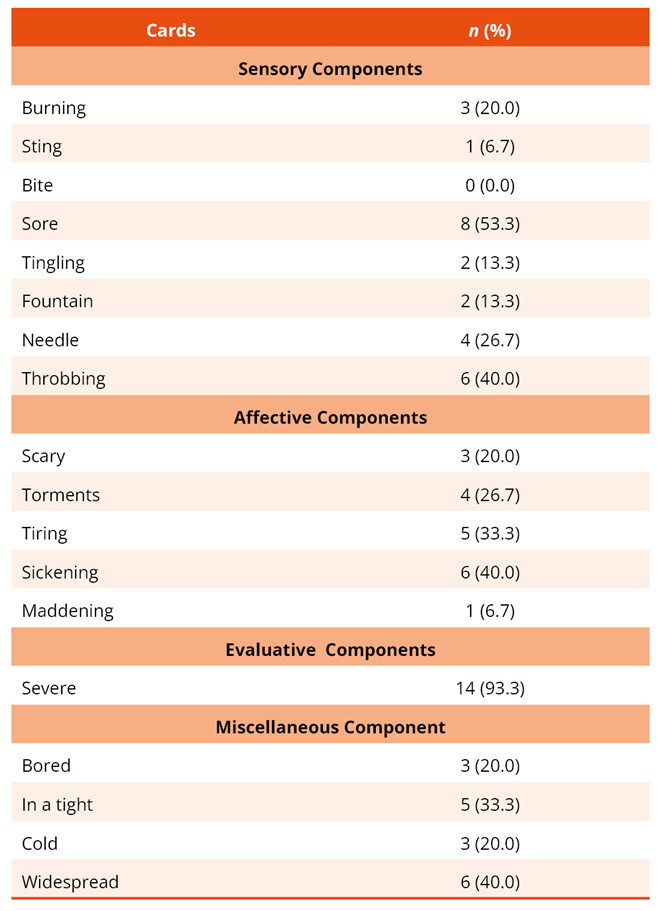
Regarding pain intensity, participants had a mean score of 6.00 with a standard deviation of 1.633 on the Wong-Baker FACES® scale. When selecting pain quality descriptor cards, 14 described their pain as severe (93.3%), 8 (53.3%) as aching, and 6 (40.0%) as throbbing, sickening, and widespread (Table 2).
Table 2. Description of pain characteristic cards chosen by the participants (n = 15)
Regarding quality of life, both children and their caregivers perceived substantial impact across all domains, particularly in social activities. However, children felt that after this domain, the most compromised aspect was emotional functioning, whereas parents identified physical functioning as the next most affected (Table 3).
Table 3. Participants' self-report and parents' report on quality of life (n = 15)
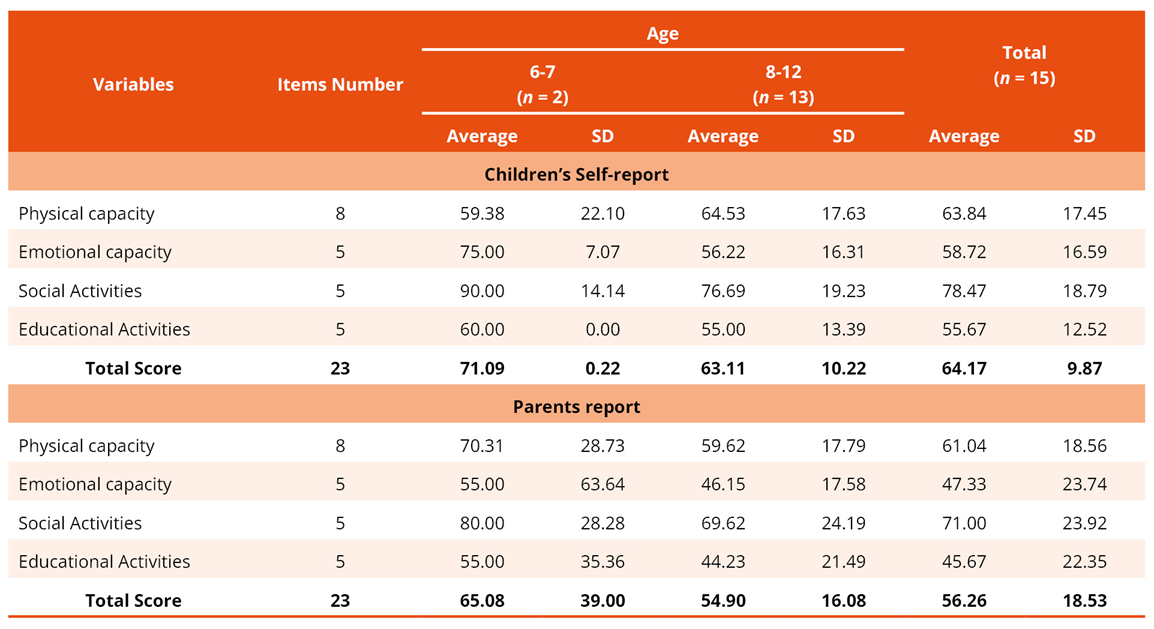
Legend: SD = Standard Deviation
Following a single session of LASER acupuncture, the average pain intensity decreased from 6.00 (SD = 1.633) to 1.75 (SD = 2.363) on the Wong-Baker FACES® scale (p = 0.036) in the intragroup analysis.
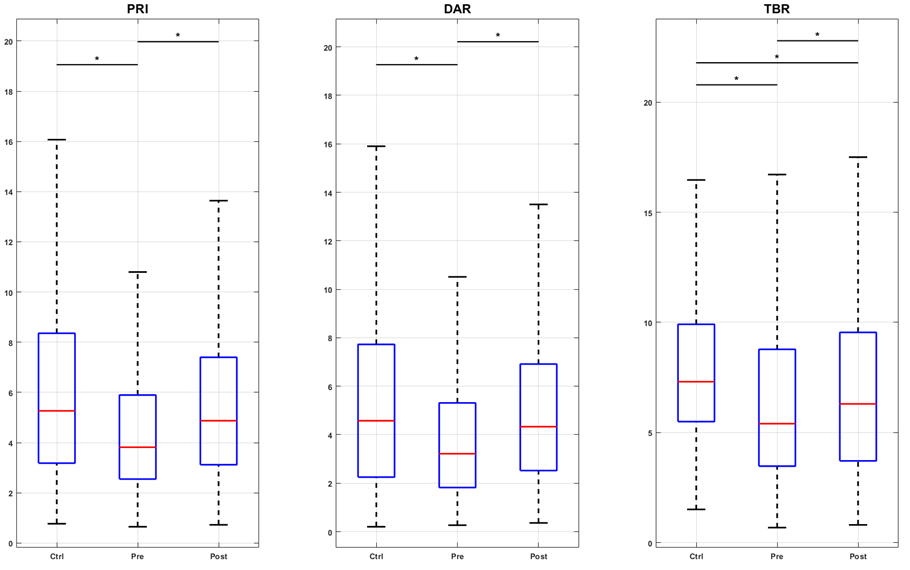
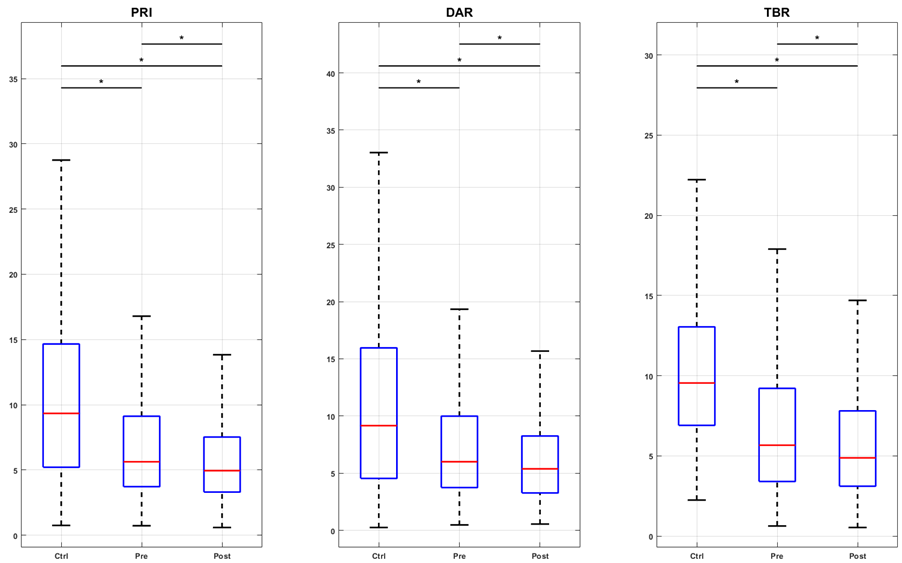
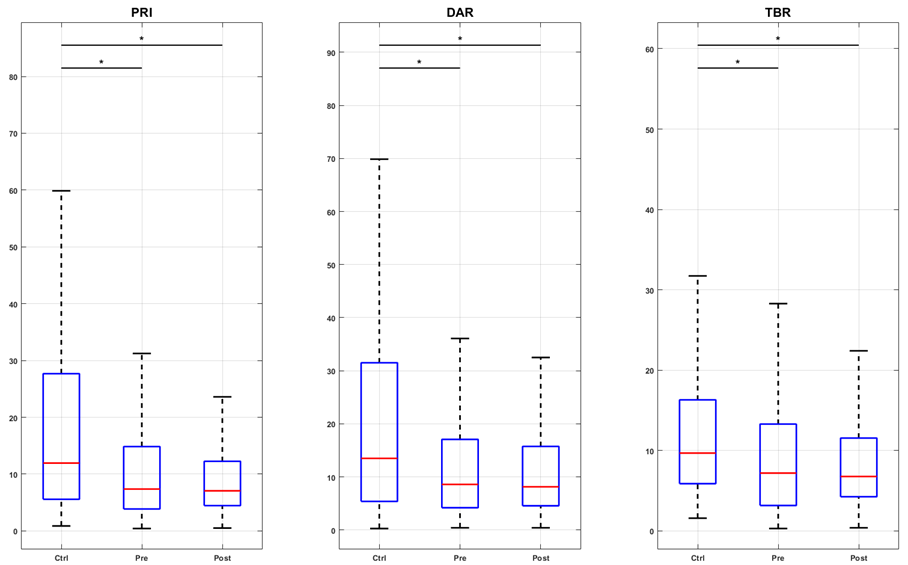
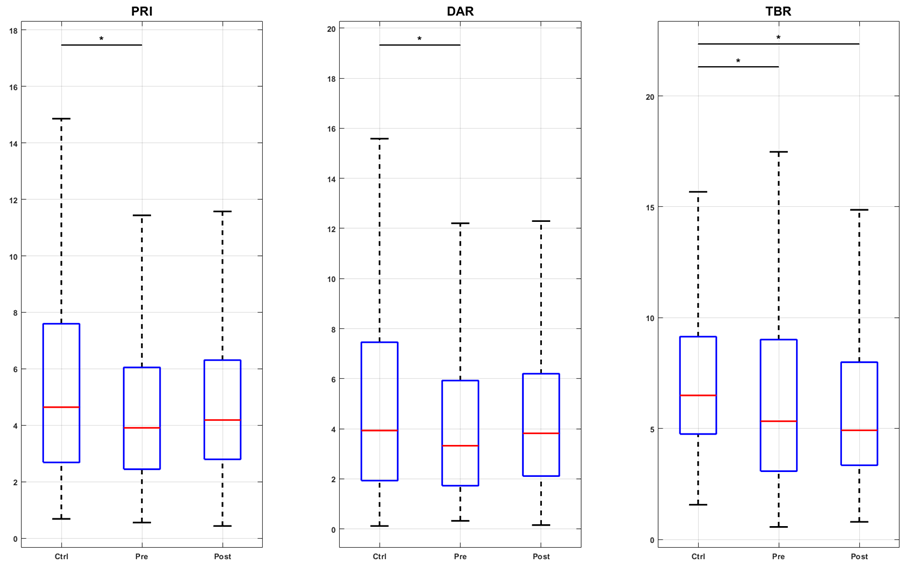
Changes in EEG tracings were observed alongside modifications in pain intensity. Figure 2a illustrates the PRI, DAR, and TBR indices for the control group (Contr G), SCD children, and the pre- and post-intervention conditions. The results indicated that LASER acupuncture successfully restored the PRI, DAR, and TBR indices in the sensory-motor region, which was associated with a slowing of EEG activity. In contrast, in the prefrontal region (F3, F4, F7, F8, Fz, Fp1, and Fp2), the intervention increased higher frequency activity, which corresponded to a decrease in the three indices below both the pre-intervention and control values (Figures 2b and 2c). For the parietal region (P3, P4, and Pz), the only significant change was the increase in theta over beta activity after LASER acupuncture (Figure 2d).
Figure 2
Figure 2a. Boxplots of spectral indexes (PRI, DAR, and TBR) values for the groups control (Ctrl), pre- and post-intervention (Pre and Post), considering the central channels C3, C4, and Cz. The inter-group analysis was performed by the Kruskal-Wallis test. Chi-square statistics: 83.09; 68.75; 106.10. Asterisk indicates a significant difference (p-value < 0.01) in the post hoc test. Figure 2b. Boxplots of spectral indexes (PRI, DAR, and TBR) values for the groups control (Ctrl), pre- and post-intervention (Pre and Post), considering the frontal channels F3, F4, F7, F8, and Fz. The inter-group analysis was performed by the Kruskal-Wallis test. Chi-square statistics: 252.37; 138.88; 449.49. Asterisk indicates a significant difference (p-value < 0.01) in the post hoc test. Figure 2c. Boxplots of spectral indexes (PRI, DAR, and TBR) values for the groups control (Ctrl), pre- and post-intervention (Pre and Post), considering the prefrontal channels FP1 and FP2. The intergroup analysis was performed using the Kruskal-Wallis test. Chi-square statistics: 123.44; 81.50; 84.16. Asterisk indicates a significant difference (p-value < 0.01) in the post hoc test. Figure 2d. Boxplots of spectral indexes (PRI, DAR, and TBR) values for the groups control (Ctrl), pre- and post-intervention (Pre and Post), considering the parietal channels P3, P4, and Pz. The inter-group analysis was performed by the Kruskal-Wallis test. Chi-square statistics: 20.58; 13.85; 76.39. Asterisk indicates a significant difference (p-value < 0.01) in the post hoc test.
Possible adverse effects were assessed during the children's participation in the study. No child reported discomfort, malaise, or any other symptoms throughout the intervention. Regarding individual protection, depending on the acupuncture point used, the use of protective glasses was recommended. Aside from this preventive measure, no other risks of harm to the physical, psychological, moral, intellectual, social, cultural, or spiritual dimensions were identified at any stage of the research, nor as a result of it.
4. Discussion
This pilot study, which aimed to verify the feasibility of using photobiomodulation through LASER acupuncture to control pain in children with SCD, identified that this therapeutic modality is safe and has a potential to reduce pain intensity, which happens together with changes in brain activity. The study demonstrates a significant reduction in high baseline pain intensity following a single session biophotobiomodulation using LASER acupuncture, without any type of adverse events. These results are not aligned with a previous meta-analysis, which showed that the analgesic effects of this type of intervention are often seen after long-term periods, and not immediately after the sessions12. Previous studies have suggested LASER acupuncture may have an immediate analgesic effect in procedural pain in children13,14. Children with SCD often display high-intensity chronic pain15, which affects both sensory and affective components. As they are often exposed to procedural pain16, maybe LASER acupuncture has been well received because of its non-painful nature, but this needs to be further investigated. The photobiomodulation through LASER acupuncture is a safe and well-tolerated procedure17, as well as EEG assessment, which was also well-tolerated and did not provoke any side effects.
Head was a pain location referred by this sample. It has been shown that high prevalence of stroke is not merely a marker of past injury but a key to understanding a common and debilitating symptom: headache. Silent ischemic strokes are a consequence of vaso-occlusion and endothelial dysfunction in the cerebral vasculature18. This ongoing vascular insult can lead to chronic cerebral hypoperfusion, altered cerebral blood flow velocity, and neuroinflammation19. Therefore, the reported headaches may likely be a direct clinical manifestation of the underlying cerebrovascular disease. They may represent symptoms of silent cerebral infarcts, transient ischemic attacks, or a chronic headache syndrome secondary to the altered neurovascular condition20, suggesting a shared pathophysiology. The central nervous system is not a passive bystander in SCD-related pain. Chronic pain from vaso-occlusive crises can lead to central sensitization. Conversely, cerebrovascular injury may disrupt descending inhibitory pain pathways, thereby lowering the threshold for and increasing the perception of pain21,22. This creates a vicious cycle where cerebral damage and chronic pain perpetuate one another.
An analysis of pain localization revealed other affected areas as the lumbosacral region, knees, and thoracic area emerging as the primary sites of complaint. This distribution aligns with the established literature on vaso-occlusive crises, where pain in the long bones of the extremities, back, and chest is highly prevalent due to marrow ischemia23. A particularly noteworthy observation, however, was the absence of the hip among the most frequently reported pain sites. This is counterintuitive, given that the hip joint is a common location for avascular necrosis of the femoral head in SCD. The most plausible explanation for this discrepancy lies in the demographic profile of our study population. In younger children and early adolescents, the femoral head is still largely cartilaginous and has a more robust collateral blood supply, rendering it less susceptible to irreversible ischemic damage24. While radiographic evidence of avascular necrosis can be present in asymptomatic children, the progression of pain and functional limitation typically manifests in older adolescents and young adults as the skeleton matures and the weight-bearing demands on the hip increase25. The lumbosacral and knee pain are classic presentations of vaso occlusive crises in marrow-rich bones, while thoracic pain may indicate acute chest syndrome26.
The characterization of participants' pain by the card-based assessment tool reveals a multidimensional experience. Descriptors indicating that pain is not merely a sensory signal but an experience shaped by cognitive, emotional, and contextual factors27. It underscores that the pain experience in SCD is deeply interwoven with emotional suffering, a finding consistently reported in qualitative studies of this population27. This affective burden can amplify the perception of pain intensity, leading to a state of hypervigilance and catastrophic thinking24. The reported compromise in the "sensory" dimension confirms the intense nociceptive and neuropathic mechanisms at play during a vaso-occlusive crisis22. However, the co-occurrence of strong sensory and affective descriptors suggests a phenomenon of central sensitization, where the central nervous system becomes hyperexcitable, amplifying both the physical sensation and the emotional distress associated with pain16. The "miscellaneous" domain, often capturing the disruptive and overwhelming nature of pain. This aligns with the concept of "total pain," which acknowledges that severe chronic and recurrent pain affects every facet of an individual's life, including their functional capacity, social interactions, and sense of self21.
EEG activity was assessed through indices that show trends of lowering or speeding electrical activity in the brain. The results are innovative regarding the assessment of brain activity in children with SCD28. As the brain develops, the preponderance of low-frequency activity (increased theta power-density) gives place to higher frequencies over the entire brain, with the cut-off occurring around 10 years old29. The preponderance of power-density ranges through indices has shown in a small sample of children with SCD that low predominated over higher EEG frequencies in their brains, a result that was opposite to ours. According to our findings, children with SCD and pain had a predominance of higher EEG frequencies over the entire brain. LASER acupuncture increased low-frequency activity and restored it to control values in the sensory-motor region, which is related to the antinociceptive pathway30. However, EEG activity sped up in the prefrontal region, which was not expected previously. The findings that PRI, DAR, and TBR decreased away from control values in the prefrontal cortex may be interpreted with caution, as the literature regarding brain electrical activity in SCD children is sparse. The PFC has an already established role in the cognitive-affective dimension of pain, as it is part of the default mode network31. Elevated DAR and TBR are known as markers of cognitive load and dysregulation in chronic pain conditions. The normalization of these rhythms may suggest a restoration of PFC function in the top-down control of pain.
The data regarding the impact of pain on quality of life reveal a critical and nuanced narrative: while both children with SCD and their caregivers converge on the significant disruption of social activities, their perceptions diverge markedly regarding the domains of physical and emotional functioning. This discordance provides a profound insight into the lived experience of pediatric chronic pain. The unanimous report of significant impairment in social activities is a stark testament to the isolating nature of SCD. Recurrent and unpredictable pain episodes inevitably lead to school absences, withdrawal from peer interactions, and an inability to participate in communal events, effectively fracturing the child's social world32. Parents witness the tangible limitations: their child's inability to walk during a crisis, their refusal to engage in play, or their visible grimacing and guarding. Their assessment is logically grounded in these overt behavioral and functional cues. In contrast, the children themselves reported a greater impact on their emotional capacity. This internal perspective highlights the profound psychological burden of SCD that may not be fully visible to an observer. Children are more acutely aware of the fear of impending pain, the frustration of missed opportunities, the sadness of isolation, and the anxiety of feeling "different" from their peers33. This finding is consistent with the literature on "internalizing" symptoms in chronic pediatric illness, where emotional distress is often masked by physical complaints. This parent-child discrepancy is not a matter of one report being more accurate than the other; rather, it reflects the multifaceted nature of suffering in SCD. The child's reality encompasses the internal emotional tsunami triggered by the pain, while the parent's focus is rightly on managing the external physical storm. This disconnect has direct clinical implications. It suggests that healthcare providers must actively and separately solicit the child's perspective on their emotional well-being, as relying solely on parent proxy-reports may lead to an underestimation of the child's psychological distress33. Comprehensive care must, therefore, integrate psychological support that addresses the internal emotional landscape described by the children themselves, in addition to managing the physical symptoms observed by their caregivers34,35.
The findings of the present study have limitations that include small sample size, the short duration of the intervention and follow-up, and the unblinded exploratory nature of the study design. This sample is not representative of the interest population just because it is a pilot study limiting the results generalization. Data about pain intensity effect were not analyzed by statistic methods to reduce risk of sub or super estimation. However, due to the nature of EEG data, neurobiological plausibility was analyzed to support the future randomized clinical trial. These findings are crucial for refining procedures, data collection instruments, and avoiding major research flaws.
In summary, our findings reveal that the LASER acupuncture promising potential for brain-mediated pain modulation in children with sickle cell disease. However, it is necessary to conduct the randomized clinical trials with high methodological accuracy to conclude about the efficacy of this new and non-invasive approach to control and prevent central pain and brain maladaptive alterations.
Use of AI
The authors declare that they used aid of ChatGPT, DeepSeek and Google Translator to find relevant articles, improve translation and writing the manuscript. However, the ideas and scientific interpretation were produced by themselves, and ultimate responsibility for the published content belong exclusively to the authors.
Funding
All costs associated with the development of the project were borne by the researchers themselves.
Authors' contributions
The authors declared that they have made substantial contributions to the work in terms of the conception or design of the research; the acquisition, analysis, or interpretation of data for the work; and the writing or critical review for relevant intellectual content. All authors approved the final version to be published and agreed to take public responsibility for all aspects of the study.
Competing interests
No financial, legal, or political conflicts involving third parties (government, private companies, and foundations, etc.) were declared for any aspect of the submitted work (including but not limited to grants and funding, advisory board participation, study design, manuscript preparation, statistical analysis, etc.).
References
1. Araújo AS, Pinto ACS, Lobo CLC, Figueiredo MS, Gualandro SFM, Saad STO, et al. Sickle Cell Disease in Brazil: Current Management. Hemoglobin. 2024;48(4):218-30. https://doi.org/10.1080/03630269.2024.2344790
2. Farrell AT, Panepinto J, Carroll CP, Darbari DS, Desai AA, King AA, et al. End points for sickle cell disease clinical trials: patient-reported outcomes, pain, and the brain. Blood Adv. 2019;3(23):3982-4001. https://doi.org/10.1182/bloodadvances.2019000882
3. Cançado RD, Jesus JA. Sickle cell disease in Brazil. Rev Bras Hematol Hemoter. 2007;29(3):204-6. https://doi.org/10.1590/S1516-84842007000300002
4. Chou ST, Alsawas M, Fasano RM, Field JJ, Hendrickson JE, Howard J, et al. American Society of Hematology 2020 guidelines for sickle cell disease: transfusion support. Blood Adv. 2020;4(2):327-55. https://doi.org/10.1182/bloodadvances.2019001143
5. Hong Y, Wu W, Wang S, Hao Q, Zheng H, Li S, et al. Angiotensin II type 1 receptor blockade attenuates posttraumatic stress disorder-related chronic pain by inhibiting glial activation in the spinal cord. Neuropharmacology. 2021;196:108704. https://doi.org/10.1016/j.neuropharm.2021.108704
6. Bender MA, Carlberg K. Sickle Cell Disease. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews®. Seattle (WA): University of Washington, Seattle; 1993-2025. Cited: PMID: 20301551
7. Connolly ME, Bills SE, Hardy SJ. Neurocognitive and psychological effects of persistent pain in pediatric sickle cell disease. Pediatr Blood Cancer. 2019;66(9):e27823. https://doi.org/10.1002/pbc.27823
8. Wang Y, Hardy SJ, Ichesco E, Zhang P, Harris RE, Darbari DS. Alteration of grey matter volume is associated with pain and quality of life in children with sickle cell disease. Transl Res. 2022;240:17-25. https://doi.org/10.1016/j.trsl.2021.08.004
9. Darbari DS, Hampson JP, Ichesco E, Kadom N, Vezina G, Evangelou I, et al. Frequency of Hospitalizations for Pain and Association With Altered Brain Network Connectivity in Sickle Cell Disease. J Pain. 2015;16(11):1077-86. https://doi.org/10.1016/j.jpain.2015.07.005
10. Estcourt LJ, Fortin PM, Hopewell S, Trivella M, Doree C, Abboud MR. Interventions for preventing silent cerebral infarcts in people with sickle cell disease. Cochrane Database Syst Rev. 2017;5(5):CD012389. https://doi.org/10.1002/14651858.CD012389.pub2
11. Law D, McDonough S, Bleakley C, Baxter GD, Tumilty S. Laser acupuncture for treating musculoskeletal pain: a systematic review with meta-analysis. J Acupunct Meridian Stud. 2015;8(1):2-16. https://doi.org/10.1016/j.jams.2014.06.015
12. MacPherson H, Altman DG, Hammerschlag R, Youping L, Taixiang W, White A, et al. Revised STandards for Reporting Interventions in Clinical Trials of Acupuncture (STRICTA): extending the CONSORT statement. PLoS Med. 2010;7(6):e1000261. https://doi.org/10.1371/journal.pmed.1000261
13. Pooja B, Kamatham R, Anchala K, Avisa P. Effectiveness of low-level laser therapy in reducing pain perception of children during dental local anesthetic administration using laser acupuncture pen on the LI4 point: a randomized clinical trial. Eur Arch Paediatr Dent. 2023;24(2):219-27. https://doi.org/10.1007/s40368-023-00780-8
14. Vögtle E, Kröner-Herwig B, Barke A. Nocebo Hyperalgesia can be Induced by the Observation of a Model Showing Natural Pain Expressions. Clin J Pain. 2019;35(9):737-43. https://doi.org/10.1097/AJP.0000000000000734
15. Machado R, Machado A, Almeida HS, Carvalho A, Sá KN. Pain in children and adolescents with falciform disease: observational study. J Physiother Res. 2021;11(2):384-92. https://doi.org/10.17267/2238-2704rpf.v11i2.3771
16. Schlenz AM, McClellan CB, Mark TR, McKelvy AD, Puffer E, Roberts CW, et al. Sensitization to acute procedural pain in pediatric sickle cell disease: modulation by painful vaso-occlusive episodes, age, and endothelin-1. J Pain. 2012;13(7):656-65. https://doi.org/10.1016/j.jpain.2012.04.001
17. Yang J, Mallory MJ, Wu Q, Bublitz SE, Do A, Xiong D, et al. The Safety of Laser Acupuncture: A Systematic Review. Med Acupunct. 2020;32(4):209-17. https://doi.org/10.1089/acu.2020.1419
18. DeBaun MR, Kirkham FJ. Central nervous system complications and management in sickle cell disease. Blood. 2016;127(7):829-38. https://doi.org/10.1182/blood-2015-09-618579
19. Verduzco LA, Nathan DG. Sickle cell disease and stroke. Blood. 2009;114(25):5117-25. https://doi.org/10.1182/blood-2009-05-220921
20. Houwing ME, Grohssteiner RL, Dremmen MHG, Atiq F, Bramer WM, de Pagter APJ, et al. Silent cerebral infarcts in patients with sickle cell disease: a systematic review and meta-analysis. BMC Med. 2020;18(1):393. https://doi.org/10.1186/s12916-020-01864-8
21. Ballas SK, Darbari DS. Review/overview of pain in sickle cell disease. Complement Ther Med. 2020;49:102327. https://doi.org/10.1016/j.ctim.2020.102327
22. Uwaezuoke SN, Ayuk AC, Ndu IK, Eneh CI, Mbanefo NR, Ezenwosu OU. Vaso-occlusive crisis in sickle cell disease: current paradigm on pain management. J Pain Res. 2018;11:3141-50. https://doi.org/10.2147/JPR.S185582
23. Gupta P, Shrivastava S, Kumar R. Musculoskeletal complications in sickle cell disease: Pathophysiology, diagnosis and management. Best Pract Res Clin Rheumatol. 2025;39(2):102033. https://doi.org/10.1016/j.berh.2025.102033
24. Gatchel RJ, McGeary DD, McGeary CA, Lippe B. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69(2):119-30. https://doi.org/10.1037/a0035514
25. Jenerette CM, Brewer CA, Ataga KI. Care seeking for pain in young adults with sickle cell disease. Pain Manag Nurs. 2014;15(1):324-30. https://doi.org/10.1016/j.pmn.2012.10.007
26. Corman M, Dambrun M, Ginzac A, Ménard K. Exploring the concept of Total Pain in contemporary oncology palliative care: a qualitative study on patients' resources. BMC Palliat Care. 2025;24(1):85. https://doi.org/10.1186/s12904-025-01719-0
27. Badawy SM, Cronin RM, Hankins J, Crosby L, DeBaun M, Thompson AA, et al. Patient-Centered eHealth Interventions for Children, Adolescents, and Adults With Sickle Cell Disease: Systematic Review. J Med Internet Res. 2018;20(7):e10940. https://doi.org/10.2196/10940
28. Cellier D, Riddle J, Petersen I, Hwang K. The development of theta and alpha neural oscillations from ages 3 to 24 years. Dev Cogn Neurosci. 2021;50:100969. https://doi.org/10.1016/j.dcn.2021.100969
29. Panepinto JA, Kato GJ, Smith WR. Health-related quality of life in sickle cell disease. Nat Rev Dis Primers. 2019;5(1):27. https://doi.org/10.1038/s41572-019-0080-5
30. Lopes TS, Ballas SK, Santana JERS, Melo-Carneiro P, Oliveira LB, Sá KN, et al. Sickle cell disease chronic joint pain: clinical assessment based on maladaptive central nervous system plasticity. Front Med. 2022;9:679053. https://doi.org/10.3389/fmed.2022.679053
31. Case M, Zhang H, Mundahl J, Datta Y, Nelson S, Gupta K, et al. Characterization of functional brain activity and connectivity using EEG and fMRI in patients with sickle cell disease. Neuroimage Clin. 2016;14:1-17. https://doi.org/10.1016/j.nicl.2016.12.024
32. Logan DE, Simons LE, Carpino EA. Too sick for school? Parent influences on school functioning among children with chronic pain. Pain. 2012;153(2):437-43. https://doi.org/10.1016/j.pain.2011.11.004
33. Roy R, de la Vega R, Jensen MP, Miró J. Neurofeedback for Pain Management: A Systematic Review. Front Neurosci. 2020;14:671. https://doi.org/10.3389/fnins.2020.00671
34. Jones EG. Thalamocortical dysrhythmia and chronic pain. Pain. 2010;150(1):4-5. https://doi.org/10.1016/j.pain.2010.03.022
35. Ploner M, Sorg C, Gross J. Brain Rhythms of Pain. Trends Cogn Sci. 2017;21(2):100-10. https://doi.org/10.1016/j.tics.2016.12.001
